24.Copolyimide Asymmetric Hollow Fiber Membranes for High-Pressure Natural Gas Purification
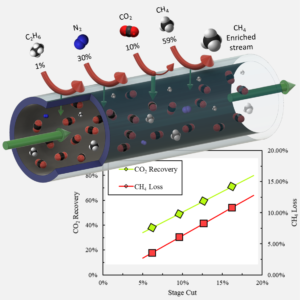 The current process to purify contaminated natural gas reserves employs a highly energy-intensive amine scrubbing technology. To satisfy the growing demand for natural gas coupled with the tight regulations on CO2 emissions, researchers worldwide are investigating the use of polymeric membranes for acid gas removal from natural gas streams, to reduce the energy consumption and carbon emission of the nowadays used technology. Placing a polymeric membrane unit at the upstream of the amine scrubbing column reduces the energy consumption during the regeneration process of saturated amines. Hence, the preparation of polymeric membranes in form of hollow fibers is a key step towards their industrial implementation for natural gas processing. The following study demonstrates the potential separation performance of 6FDA-Durene/6FDA-CARDO (5000:5000) in an integrally skinned defect-free asymmetric hollow fiber geometry. The initial screening studies of the membrane shows good pure-gas performance as it exhibits CO2/CH4 selectivity coefficients around 28 and CO2 permeance between 310 and 350 GPU. Feed pressures of up to 900 psig and stage cuts ranging from 5% to 20% were tested for a quaternary sweet mixed-gas feed to assess the membrane performance under operating conditions that mimic those in industrial settings. Mixed-gas permeance of all gases, and CO2/CH4 selectivity remain somewhat constant as the feed pressure increases up to 900 psig. Both mixed-gas CO2 permeance and CO2/CH4 selectivity decline as the stage cut increases. As expected, as CO2 recovery is maximized, CO2 purity is minimized and CH4 loss increases with increasing stage cut. As a result of the trade-off between recovery and purity, the data and analyses reported herein can provide insights into the limitations that certain operating parameters can pose on CO2 separation performance with similar polymeric hollow fiber membranes.
The current process to purify contaminated natural gas reserves employs a highly energy-intensive amine scrubbing technology. To satisfy the growing demand for natural gas coupled with the tight regulations on CO2 emissions, researchers worldwide are investigating the use of polymeric membranes for acid gas removal from natural gas streams, to reduce the energy consumption and carbon emission of the nowadays used technology. Placing a polymeric membrane unit at the upstream of the amine scrubbing column reduces the energy consumption during the regeneration process of saturated amines. Hence, the preparation of polymeric membranes in form of hollow fibers is a key step towards their industrial implementation for natural gas processing. The following study demonstrates the potential separation performance of 6FDA-Durene/6FDA-CARDO (5000:5000) in an integrally skinned defect-free asymmetric hollow fiber geometry. The initial screening studies of the membrane shows good pure-gas performance as it exhibits CO2/CH4 selectivity coefficients around 28 and CO2 permeance between 310 and 350 GPU. Feed pressures of up to 900 psig and stage cuts ranging from 5% to 20% were tested for a quaternary sweet mixed-gas feed to assess the membrane performance under operating conditions that mimic those in industrial settings. Mixed-gas permeance of all gases, and CO2/CH4 selectivity remain somewhat constant as the feed pressure increases up to 900 psig. Both mixed-gas CO2 permeance and CO2/CH4 selectivity decline as the stage cut increases. As expected, as CO2 recovery is maximized, CO2 purity is minimized and CH4 loss increases with increasing stage cut. As a result of the trade-off between recovery and purity, the data and analyses reported herein can provide insights into the limitations that certain operating parameters can pose on CO2 separation performance with similar polymeric hollow fiber membranes.
Shalabi, Y.A.; Yahaya, G.O.; Choi, S.-H.; Alsamah, A.; Hayek, A.; Journal of Applied Polymer Science, 2023, xxx, 53866. DOI: 10.1002/app.53866.
23.Modified CARDO-Based Copolyimides with Improved Sour Mixed-Gas Permeation Properties
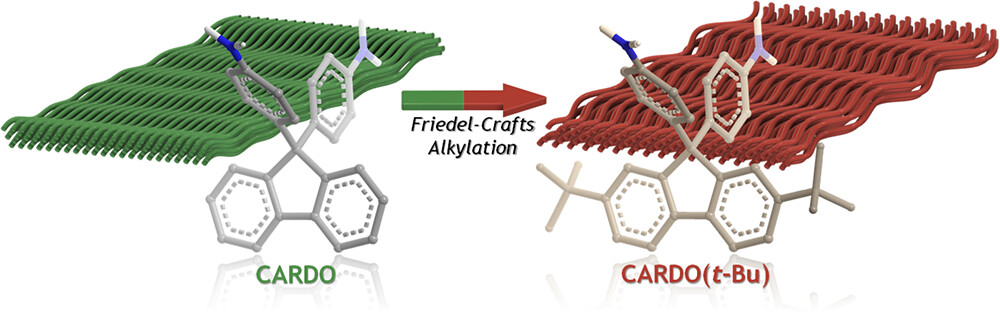 Chemical modification is an interesting tool to introduce functional groups into polymers’ backbones to tailor their properties in a desired fashion. Polymers used in membrane-based gas separation applications need to possess high gas permeability and selectivity during mixed-gas separation under harsh operational conditions of pressure and temperature. Herein, we report the modification of three copolyimides containing the moiety 9,9-bis(4-aminophenyl)fluorine (CARDO) through Friedel–Crafts alkylation to introduce bulky tert-butyl groups into their backbones. Membranes prepared from the modified polymers were evaluated using pure- and sweet mixed-gas under aggressive feed pressures. Based on the promising results obtained, a selected copolyimide was subjected to sour mixed-gas separation under feed pressures up to 500 psi. Mixed-gas separation studies, especially those containing hydrogen sulfide, are of great interest to develop membranes for use in sour natural gas reserve treatment. For example, the H2S and CO2 permeability coefficients of 6FDA-Durene/6FDA-CARDO(t-Bu) at 500 psi were recorded as 456 and 238 Barrer, respectively, where the H2S/CH4 and CO2/CH4 are 20.4 and 10.6, respectively. The obtained results are very promising and even superior to many glassy polymers reported previously. This work represents an example of the benefit of chemical modification on polymers to tailor their structure–property relationship. Future works will be focused on the fabrication and testing of asymmetric or composite hollow fibers to imitate the membrane modules used industrially.
Chemical modification is an interesting tool to introduce functional groups into polymers’ backbones to tailor their properties in a desired fashion. Polymers used in membrane-based gas separation applications need to possess high gas permeability and selectivity during mixed-gas separation under harsh operational conditions of pressure and temperature. Herein, we report the modification of three copolyimides containing the moiety 9,9-bis(4-aminophenyl)fluorine (CARDO) through Friedel–Crafts alkylation to introduce bulky tert-butyl groups into their backbones. Membranes prepared from the modified polymers were evaluated using pure- and sweet mixed-gas under aggressive feed pressures. Based on the promising results obtained, a selected copolyimide was subjected to sour mixed-gas separation under feed pressures up to 500 psi. Mixed-gas separation studies, especially those containing hydrogen sulfide, are of great interest to develop membranes for use in sour natural gas reserve treatment. For example, the H2S and CO2 permeability coefficients of 6FDA-Durene/6FDA-CARDO(t-Bu) at 500 psi were recorded as 456 and 238 Barrer, respectively, where the H2S/CH4 and CO2/CH4 are 20.4 and 10.6, respectively. The obtained results are very promising and even superior to many glassy polymers reported previously. This work represents an example of the benefit of chemical modification on polymers to tailor their structure–property relationship. Future works will be focused on the fabrication and testing of asymmetric or composite hollow fibers to imitate the membrane modules used industrially.
Hayek, A.; Alsamah, A.; Saleem, Q.; Alhajry, R.H.; Alsuwailem, A.A.; Jassim, F.I. ACS Appl. Polym. Mater. 2022, 4, 12, 9257–9271. DOI: 10.1021/acsapm.2c01527.
22.Molecular Design of Poly(imide-oxadiazole) Membranes for High-Pressure Mixed-Gas Separation
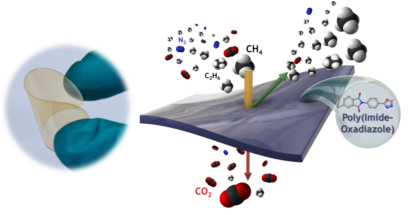 The design of new polymeric materials and the search for new classes of polymers for industrial application in membrane-based gas separation have been the focus of several research groups worldwide. Membranes with high productivity and efficiency under harsh operational conditions of pressure and temperature during mixed-gas separation are of great interest. In this paper, we report the preparation of a series of block copolymers of poly(imide–oxadiazole)s built from 6FDA, Durene, and four different 1,3,4-oxadiazole-based monomers. The pure- and mixed-gas separation properties of their corresponding membranes were measured. The mixed-gas separation data were collected at a high feed pressure of up to 900 psi. Such mixed-gas separation testing under harsh conditions is required to subject the membranes to environments similar to industrial use. For example, the mixed-gas CO2 permeability of 6FDA-Durene/6FDA-4BAO(Me) (3:1) was measured at 900 psi to be ∼130 barrer, and the CO2/CH4 selectivity is around ∼24. These results of permeability and selectivity of poly(imide–oxadiazole) membranes at such high gas feed pressure are considered very attractive and are superior to many glassy polymers reported in the literature and that are industrially observed membranes. This work illustrates well a case scenario of structure–property relationship and demonstrates the exclusivity of mixed-gas testing which could not be predicted from the ideal situation of pure-gas testing. In the near future, the mixed-gas separation properties of thin layer composite and hollow fibers will be evaluated.
The design of new polymeric materials and the search for new classes of polymers for industrial application in membrane-based gas separation have been the focus of several research groups worldwide. Membranes with high productivity and efficiency under harsh operational conditions of pressure and temperature during mixed-gas separation are of great interest. In this paper, we report the preparation of a series of block copolymers of poly(imide–oxadiazole)s built from 6FDA, Durene, and four different 1,3,4-oxadiazole-based monomers. The pure- and mixed-gas separation properties of their corresponding membranes were measured. The mixed-gas separation data were collected at a high feed pressure of up to 900 psi. Such mixed-gas separation testing under harsh conditions is required to subject the membranes to environments similar to industrial use. For example, the mixed-gas CO2 permeability of 6FDA-Durene/6FDA-4BAO(Me) (3:1) was measured at 900 psi to be ∼130 barrer, and the CO2/CH4 selectivity is around ∼24. These results of permeability and selectivity of poly(imide–oxadiazole) membranes at such high gas feed pressure are considered very attractive and are superior to many glassy polymers reported in the literature and that are industrially observed membranes. This work illustrates well a case scenario of structure–property relationship and demonstrates the exclusivity of mixed-gas testing which could not be predicted from the ideal situation of pure-gas testing. In the near future, the mixed-gas separation properties of thin layer composite and hollow fibers will be evaluated.
Hayek, A.; Alsamah, A.; Shalabi, Y.A.; Saleem, Q.; Ben Sultan, M.M.; Alhajry, R.H.; Macromolecules 2022, 55, 9, 3747-3761. DOI: 10.1021/acs.macromol.2c00650.
21.Sour Mixed-Gas Upper Bounds of Glassy Polymeric Membranes
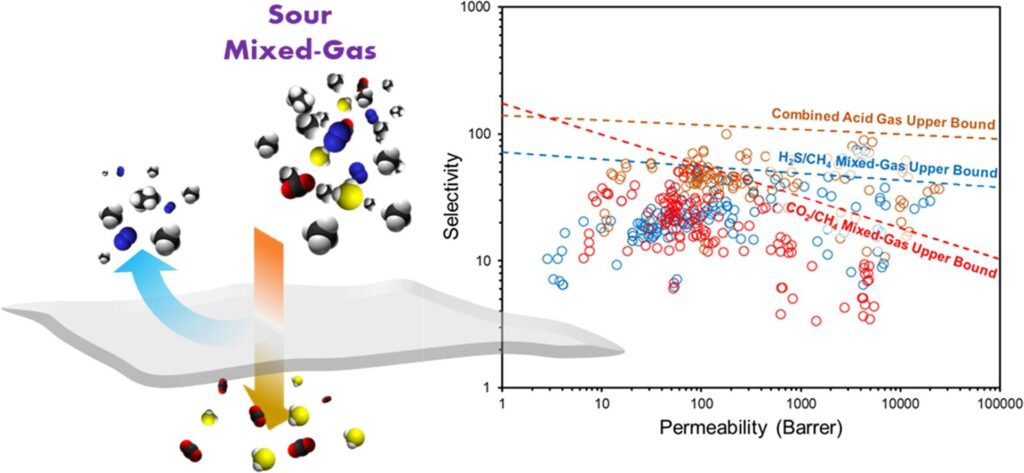 Polymeric membranes prepared from glassy polymers suffer from a permeability-selectivity trade-off relationship. When their permeability is increased, their selectivity decreases, and vice versa. The performance of membranes during pure-gas permeation is assessed by plotting their permeability and selectivity data on a corresponding pure-gas upper bound graph reported in literature. For potential use in industrial applications, it is recommended to assess the performance of membranes through their mixed-gas separation performance, as the use of pure-gas upper bounds could be misleading. As a result of this, theoretical (2015) and experimental (2018) CO2/CH4 sweet mixed-gas upper bounds have been proposed. Sour mixed-gas separation is even more complex, due to the presence of hydrogen sulfide (H2S) in the gas stream. Several H2S/CH4 trade-off lines were reported in literature based on the performance of specific materials prepared, however, in each case, the empirical parameters were drastically different. In this paper, we revisited the 2018 CO2/CH4 mixed-gas upper bound by including data measured in our laboratory and the latest sweet mixed-gas permeation data reported to date. Moreover, we propose new H2S/CH4, CO2/CH4, and combined acid gas sour mixed-gas upper bounds based on all of the sour mixed-gas permeation data reported by our research group and those available in literature. These upper bounds will serve as unique qualitative standards to design and assess new polymeric membranes developed in the future with potential use in industrial applications.
Polymeric membranes prepared from glassy polymers suffer from a permeability-selectivity trade-off relationship. When their permeability is increased, their selectivity decreases, and vice versa. The performance of membranes during pure-gas permeation is assessed by plotting their permeability and selectivity data on a corresponding pure-gas upper bound graph reported in literature. For potential use in industrial applications, it is recommended to assess the performance of membranes through their mixed-gas separation performance, as the use of pure-gas upper bounds could be misleading. As a result of this, theoretical (2015) and experimental (2018) CO2/CH4 sweet mixed-gas upper bounds have been proposed. Sour mixed-gas separation is even more complex, due to the presence of hydrogen sulfide (H2S) in the gas stream. Several H2S/CH4 trade-off lines were reported in literature based on the performance of specific materials prepared, however, in each case, the empirical parameters were drastically different. In this paper, we revisited the 2018 CO2/CH4 mixed-gas upper bound by including data measured in our laboratory and the latest sweet mixed-gas permeation data reported to date. Moreover, we propose new H2S/CH4, CO2/CH4, and combined acid gas sour mixed-gas upper bounds based on all of the sour mixed-gas permeation data reported by our research group and those available in literature. These upper bounds will serve as unique qualitative standards to design and assess new polymeric membranes developed in the future with potential use in industrial applications.
Hayek, A.; Shalabi, Y.A.; Alsamah, A.; Separation & Purification Technology, 2021, 277, 119535. DOI:10.1016/j.seppur.2021.119535.
20.Copolyimide Membranes with Improved H2S/CH4 Selectivity for High-Pressure Sour Mixed-Gas Separation
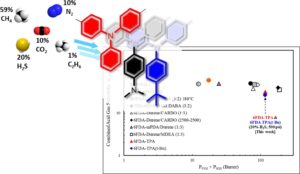
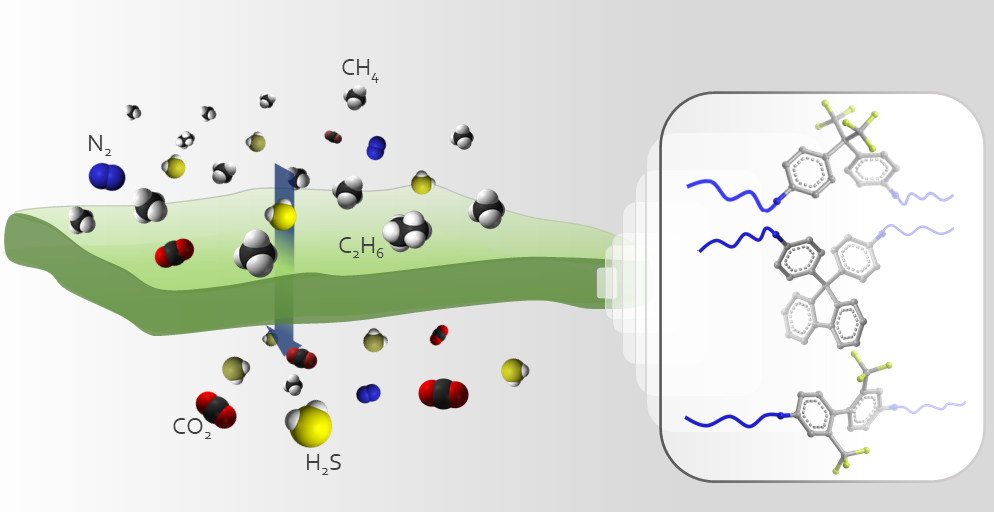 Natural gas purification is a challenging task due to the existing number of contaminants in the mixture. Sour natural gas, in particular, presents an even higher challenge due to the presence of the highly toxic hydrogen sulfide (H2S) gas. These contaminants level must be reduced to meet the sales gas specifications. In this paper, three DAM-based random copolymers were prepared for simultaneous improvement of the acid gases (CO2 and H2S) separation properties of their corresponding membranes. While the developed membranes exhibit high CO2/CH4 selectivity in a non-sour mixed-gas, their unconventional behavior during sour gas separation is difficult to predict. Therefore, their membranes were subjected to sour mixed-gas separation tests a quinary gas mixture including 20 vol% H2S at different upstream pressures up to 34.5 bar. Each membrane demonstrated one or more improved properties in terms of acid gases permeability and selectivity coefficients. Taking into consideration the overall performance, membranes prepared from 6FDA-DAM/6FpDA (1:3) exhibited the most attractive separation properties among the series prepared. However, based on the relatively high H2S/CH4 selectivity obtained of 6FDA-DAM/CARDO (1:3) membranes, a block 6FDA-DAM/6FDA-CARDO (1:1) copolyimide was prepared, and the sour mixed-gas separation properties were evaluated. The overall block copolymer membrane demonstrated very attractive productivity and efficiency, compared to the standard industrially used glassy polymer, cellulose acetate, thereby rendering it a potential candidate for sour natural gas purification.
Natural gas purification is a challenging task due to the existing number of contaminants in the mixture. Sour natural gas, in particular, presents an even higher challenge due to the presence of the highly toxic hydrogen sulfide (H2S) gas. These contaminants level must be reduced to meet the sales gas specifications. In this paper, three DAM-based random copolymers were prepared for simultaneous improvement of the acid gases (CO2 and H2S) separation properties of their corresponding membranes. While the developed membranes exhibit high CO2/CH4 selectivity in a non-sour mixed-gas, their unconventional behavior during sour gas separation is difficult to predict. Therefore, their membranes were subjected to sour mixed-gas separation tests a quinary gas mixture including 20 vol% H2S at different upstream pressures up to 34.5 bar. Each membrane demonstrated one or more improved properties in terms of acid gases permeability and selectivity coefficients. Taking into consideration the overall performance, membranes prepared from 6FDA-DAM/6FpDA (1:3) exhibited the most attractive separation properties among the series prepared. However, based on the relatively high H2S/CH4 selectivity obtained of 6FDA-DAM/CARDO (1:3) membranes, a block 6FDA-DAM/6FDA-CARDO (1:1) copolyimide was prepared, and the sour mixed-gas separation properties were evaluated. The overall block copolymer membrane demonstrated very attractive productivity and efficiency, compared to the standard industrially used glassy polymer, cellulose acetate, thereby rendering it a potential candidate for sour natural gas purification.
Yahaya, G.O.; Hayek, A.; Alsamah, A.; Shalabi, Y.A.; Ben Sultan, M.M.; Alhajry, R.H.; Separation & Purification Technology, 2021, 272, 118897. DOI:10.1016/j.seppur.2021.118897.
19. Post-Synthetic Modification of CARDO-Based Materials: Application in Sour Natural Gas Separation
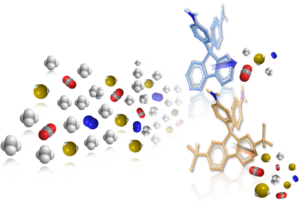 A new homopolymer, 6FDA-CARDO(t-Bu), was prepared from 6FDA-CARDO through one-step chemical modification by adding bulky tert-butyl groups, using Friedel-Crafts alkylation. The incorporation of the bulky groups led to a significant increase in the fractional free volume (FFV) within the matrix of membranes prepared from the modified polymer. The thermal properties of the modified polymer were not greatly affected compared to those of its parent 6FDA-CARDO homopolymer, where the Td5% and the Tg were measured to be 517 ºC and 366 ºC, respectively. The hydrazine-assisted cleavage of 6FDA-CARDO(t-Bu) led to the preparation of the new diamine monomer CARDO(t-Bu) in high yield. Pure-gas, and multicomponent sweet and sour mixed-gas permeation studies of dense films prepared from 6FDA-CARDO(t-Bu) were measured and compared to those of membranes prepared from 6FDA-CARDO. For example, the carbon dioxide (CO2) pure-gas permeability of 6FDA-CARDO(t-Bu) membrane was measured at 100 psi feed pressure and 22 ºC, and found to be 271 Barrer which is 4.6-fold higher when compared to that of 6FDA-CARDO membranes. Due to a simultaneous increase in the methane (CH4) pure-gas permeability; a reduction on the CO2/CH4 selectivity for 6FDA-CARDO(t-Bu) membrane was recorded. Similar effect was observed for the sweet mixed-gas separation studies. Interestingly, the sour mixed-gas (containing 21% hydrogen sulfide, H2S) separation properties of 6FDA-CARDO(t-Bu) membrane showed different trend. At 500 psi and 22 ºC, the H2S/CH4 selectivity was 10% higher, and the CO2 and H2S permeability coefficients were 3.2- and 3.8-fold, respectively, compared to those obtained for 6FDA-CARDO membranes.
A new homopolymer, 6FDA-CARDO(t-Bu), was prepared from 6FDA-CARDO through one-step chemical modification by adding bulky tert-butyl groups, using Friedel-Crafts alkylation. The incorporation of the bulky groups led to a significant increase in the fractional free volume (FFV) within the matrix of membranes prepared from the modified polymer. The thermal properties of the modified polymer were not greatly affected compared to those of its parent 6FDA-CARDO homopolymer, where the Td5% and the Tg were measured to be 517 ºC and 366 ºC, respectively. The hydrazine-assisted cleavage of 6FDA-CARDO(t-Bu) led to the preparation of the new diamine monomer CARDO(t-Bu) in high yield. Pure-gas, and multicomponent sweet and sour mixed-gas permeation studies of dense films prepared from 6FDA-CARDO(t-Bu) were measured and compared to those of membranes prepared from 6FDA-CARDO. For example, the carbon dioxide (CO2) pure-gas permeability of 6FDA-CARDO(t-Bu) membrane was measured at 100 psi feed pressure and 22 ºC, and found to be 271 Barrer which is 4.6-fold higher when compared to that of 6FDA-CARDO membranes. Due to a simultaneous increase in the methane (CH4) pure-gas permeability; a reduction on the CO2/CH4 selectivity for 6FDA-CARDO(t-Bu) membrane was recorded. Similar effect was observed for the sweet mixed-gas separation studies. Interestingly, the sour mixed-gas (containing 21% hydrogen sulfide, H2S) separation properties of 6FDA-CARDO(t-Bu) membrane showed different trend. At 500 psi and 22 ºC, the H2S/CH4 selectivity was 10% higher, and the CO2 and H2S permeability coefficients were 3.2- and 3.8-fold, respectively, compared to those obtained for 6FDA-CARDO membranes.
Hayek, A.; Alsamah, A.; Yahaya, G.O.; Qasem, E.A., Alhajry, R.H.; Journal of Materials Chemistry A, 2020, 8 (44), 23354–23367. DOI:10.1039/D0TA06967A. (This article is part of the themed collection: Journal of Materials Chemistry A HOT Papers).
18. Unprecedented Sour Mixed-Gas Permeation Properties of Fluorinated Polyazole-Based Membranes
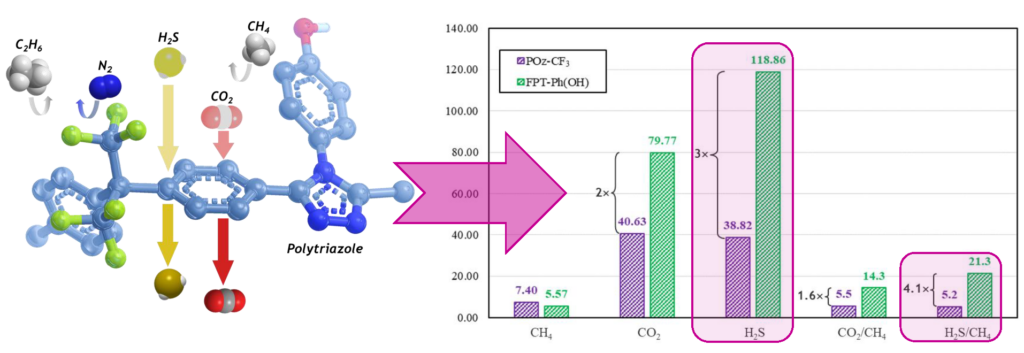 A polyoxadiazole polymer POz-CF3 was prepared, and complete gas permeation properties were investigated on its corresponding membrane using pure-, and multicomponent mixed-gas feeds. The POz-CF3 membrane demonstrated attractive pure- and sweet mixed-gas permeation properties; however, the performance of its membrane declined dramatically during sour mixed-gas testing. The modification of POz-CF3 into its corresponding polytriazole polymers led to changes in their membranes’ mechanical, thermal, processability, and gas permeation properties. For example, the membrane formation of FPT-Ph(NMe2) polytriazole could not be possible due to the polymer low solubility in most solvents. In contrast, the polytriazole FPT-Ph(OH) afforded membranes with improved mechanical properties and enhanced pure- and mixed-gas permeation properties compared to POz-CF3 membrane. The sour mixed-gas results, in particular, are very attractive and superior to most of the aromatic polymeric membranes tested with similar conditions of gas feed (20 vol % H2S) and operational pressure and temperature.
A polyoxadiazole polymer POz-CF3 was prepared, and complete gas permeation properties were investigated on its corresponding membrane using pure-, and multicomponent mixed-gas feeds. The POz-CF3 membrane demonstrated attractive pure- and sweet mixed-gas permeation properties; however, the performance of its membrane declined dramatically during sour mixed-gas testing. The modification of POz-CF3 into its corresponding polytriazole polymers led to changes in their membranes’ mechanical, thermal, processability, and gas permeation properties. For example, the membrane formation of FPT-Ph(NMe2) polytriazole could not be possible due to the polymer low solubility in most solvents. In contrast, the polytriazole FPT-Ph(OH) afforded membranes with improved mechanical properties and enhanced pure- and mixed-gas permeation properties compared to POz-CF3 membrane. The sour mixed-gas results, in particular, are very attractive and superior to most of the aromatic polymeric membranes tested with similar conditions of gas feed (20 vol % H2S) and operational pressure and temperature.
Hayek, A.; Alsamah, A.; Alaslai, N.; Maab, H.; Qasem, E.A., Alhajry, R.H.; Alyami, N.M.; ACS Applied Polymer Materials, 2020, 2(6), 2199 – 2210. DOI: 10.1021/acsapm.0c00196.
17. Development of Thin-Film Composite Membranes from Aromatic Cardo-Type Co-Polyimide for Mixed and Sour Gas Separations from Natural Gas
 The consumption of natural gas (NG) is rapidly increasing worldwide as it is becoming the second largest fuel source after coal. However, many of the world gas reserves contain high levels of subquality NG including the presence of carbon dioxide (CO2), hydrogen sulfide (H2S), nitrogen (N2), benzene/toluene/xylene (BTX) etc., in varying amounts (up to 50% v/v in some reserves), which constitute several problems. Membrane-based NG sweetening/upgrading processes emerge as among the fastest growing technologies, due to their lower capital cost, higher energy savings, greater economic viability, etc. as compared to conventional technologies. Thus, a defective-free, multilayer thin-film composite membrane is developed from 6FDA-Durene/6FDA-CARDO block co-polyimide for the separation of sour gas from NG. The membrane shows good performance as it exhibit CO2/CH4 and H2S/CH4 selectivities ranges from 8 to 10 and 15 to 19, respectively, and CO2 and H2S permeance are 122 and 220 GPU, respectively.
The consumption of natural gas (NG) is rapidly increasing worldwide as it is becoming the second largest fuel source after coal. However, many of the world gas reserves contain high levels of subquality NG including the presence of carbon dioxide (CO2), hydrogen sulfide (H2S), nitrogen (N2), benzene/toluene/xylene (BTX) etc., in varying amounts (up to 50% v/v in some reserves), which constitute several problems. Membrane-based NG sweetening/upgrading processes emerge as among the fastest growing technologies, due to their lower capital cost, higher energy savings, greater economic viability, etc. as compared to conventional technologies. Thus, a defective-free, multilayer thin-film composite membrane is developed from 6FDA-Durene/6FDA-CARDO block co-polyimide for the separation of sour gas from NG. The membrane shows good performance as it exhibit CO2/CH4 and H2S/CH4 selectivities ranges from 8 to 10 and 15 to 19, respectively, and CO2 and H2S permeance are 122 and 220 GPU, respectively.
Yahaya, G.O.; Choi, S.-H.; Sultan, M.M.B.; Hayek, A.; Global Challenges, 2020, 1900107, 1 – 8. DOI: 10.1002/gch2.201900107.
16. Fluorinated copolyimide membranes for sour mixed-gas upgrading.
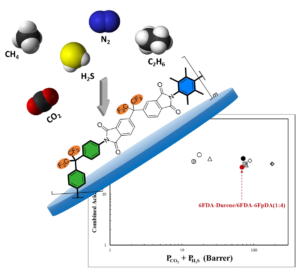 Three random and two block 4,4′‐(hexafluoroisopropylidene)diphth alic anhydride (6FDA)‐based copolyimides with different 6FpDA:Durene molar ratio varying from 25 to 80% were prepared and characterized. The pure‐gas permeation data of their membranes were investigated at 100 psi and 22 °C. The CO2/CH4 ideal selectivity coefficient increased to around 47 with the increase of the 6FpDA content to 80% in the copolymer backbone while the CO2 permeability coefficient found to be the highest (378 barrer) with highest Durene content copolymer. Based on its attractive pure‐gas permeation properties(CO2/CH4 = 47), 6FDA‐6FpDA/6FDA‐Durene (4:1) block copolyimide was selected for further analyses, where the effect of pressure and temperature on its gas transport properties was evaluated. Furthermore, the mixed‐gas permeation properties were investigated using multicomponent sweet and sour gas mixtures prepared from N2(30% or 10%), CH4 (59%), C2H6 (1%), CO2 (10%), and H2S (0% or 20%)accordingly. The sweet mixed‐gas CO2/CH4 selectivity and CO2 permeability coefficients of 6FDA‐6FpDA/6FDA‐Durene (4:1) are around 39 and 45 barrer, respectively, at elevated pressure (800 psi). The polymer, however, showed nonideal behavior when subjected to high H2S‐content gas mixture (20 vol. % H2S), where the CO2/CH4 selectivity value dropped to around 21 and the H2S/CH4 selectivity coefficient is 13. The CO2 and H2S permeability coefficients are 42 and 26 barrer, respectively, at an upstream pressure up to 500 psi. When plotted on the combined acid gas permeability‐selectivity curve, the polymer separation efficiency was nearby the high‐performing polymers reported in the literature, and way superior to the industrial standard glassy polymer, cellulose acetate, used currently in gas separation.
Three random and two block 4,4′‐(hexafluoroisopropylidene)diphth alic anhydride (6FDA)‐based copolyimides with different 6FpDA:Durene molar ratio varying from 25 to 80% were prepared and characterized. The pure‐gas permeation data of their membranes were investigated at 100 psi and 22 °C. The CO2/CH4 ideal selectivity coefficient increased to around 47 with the increase of the 6FpDA content to 80% in the copolymer backbone while the CO2 permeability coefficient found to be the highest (378 barrer) with highest Durene content copolymer. Based on its attractive pure‐gas permeation properties(CO2/CH4 = 47), 6FDA‐6FpDA/6FDA‐Durene (4:1) block copolyimide was selected for further analyses, where the effect of pressure and temperature on its gas transport properties was evaluated. Furthermore, the mixed‐gas permeation properties were investigated using multicomponent sweet and sour gas mixtures prepared from N2(30% or 10%), CH4 (59%), C2H6 (1%), CO2 (10%), and H2S (0% or 20%)accordingly. The sweet mixed‐gas CO2/CH4 selectivity and CO2 permeability coefficients of 6FDA‐6FpDA/6FDA‐Durene (4:1) are around 39 and 45 barrer, respectively, at elevated pressure (800 psi). The polymer, however, showed nonideal behavior when subjected to high H2S‐content gas mixture (20 vol. % H2S), where the CO2/CH4 selectivity value dropped to around 21 and the H2S/CH4 selectivity coefficient is 13. The CO2 and H2S permeability coefficients are 42 and 26 barrer, respectively, at an upstream pressure up to 500 psi. When plotted on the combined acid gas permeability‐selectivity curve, the polymer separation efficiency was nearby the high‐performing polymers reported in the literature, and way superior to the industrial standard glassy polymer, cellulose acetate, used currently in gas separation.
Hayek, A.; Yahaya, G.O.; Alsamah, A.; Panda, S. K.; Journal of Applied Polymer Science, 2020, 137, 48336. DOI: 10.1002/app.48336.
15. Effect of pendent bulky groups on pure- and sour mixed-gas permeation properties of triphenylamine-based polyamides.
 Three diamine triphenylamine-based monomers, 4,4′-diaminotriphenylamine (TPA), 4,4′-diamino-4″-(dimethylamino)triphenylamine (TPA(NMe2)), and 4,4′-diamino-4″-(tert-butyl)triphenylamine (TPA(t-Bu)) were reproduced in preparation for their 6FDA-based homopolyimides: 6FDA-TPA, 6FDA-TPA(NMe2) and 6FDA-TPA(t-Bu). The bulkiness effect of two pendent groups (dimethylamino and tert-butyl) on the gas transport properties of the polymers was evaluated. Both bulky groups enhanced the fractional free volume (FFV) within the membranes matrices, thereby increasing gas diffusivity, which resulted in greater CO2 permeability of 6FDA-TPA(NMe2) by almost 32% and 6FDA-TPA(t-Bu) by 3 folds compared to their nonfunctionalized analog 6FDA-TPA. Moreover, introducing bulky groups had minimal effect on gas solubility, indicating that separation through these materials is a diffusion-driven process. Afterwards, sweet mixed-gas permeation properties where measured for 6FDA-TPA and 6FDA-TPA(t-Bu) membranes using a multicomponent gas mixture (N2, CH4, C2H6 and CO2) at various feed pressures up to 500 psi and a fixed temperature of 22 °C. Once again, the introduction of the bulky group (t-Bu) resulted in an increase in the mixed-gas CO2 permeability coefficient by 3 folds, while the CO2/CH4 selectivity coefficient dropped by around 33%. Moreover, the sour mixed-gas permeation properties using a high H2S-content multicomponent gas mixture (20 vol% H2S) were measured for 6FDA-TPA and 6FDA-TPA(t-Bu) membranes at feed pressures up to 500 psi and 22 °C. The membranes showed nonideal behavior in the presence of hydrogen sulfide, where both membranes performed similarly and the effect of the bulky group (t-Bu) was negligible.
Three diamine triphenylamine-based monomers, 4,4′-diaminotriphenylamine (TPA), 4,4′-diamino-4″-(dimethylamino)triphenylamine (TPA(NMe2)), and 4,4′-diamino-4″-(tert-butyl)triphenylamine (TPA(t-Bu)) were reproduced in preparation for their 6FDA-based homopolyimides: 6FDA-TPA, 6FDA-TPA(NMe2) and 6FDA-TPA(t-Bu). The bulkiness effect of two pendent groups (dimethylamino and tert-butyl) on the gas transport properties of the polymers was evaluated. Both bulky groups enhanced the fractional free volume (FFV) within the membranes matrices, thereby increasing gas diffusivity, which resulted in greater CO2 permeability of 6FDA-TPA(NMe2) by almost 32% and 6FDA-TPA(t-Bu) by 3 folds compared to their nonfunctionalized analog 6FDA-TPA. Moreover, introducing bulky groups had minimal effect on gas solubility, indicating that separation through these materials is a diffusion-driven process. Afterwards, sweet mixed-gas permeation properties where measured for 6FDA-TPA and 6FDA-TPA(t-Bu) membranes using a multicomponent gas mixture (N2, CH4, C2H6 and CO2) at various feed pressures up to 500 psi and a fixed temperature of 22 °C. Once again, the introduction of the bulky group (t-Bu) resulted in an increase in the mixed-gas CO2 permeability coefficient by 3 folds, while the CO2/CH4 selectivity coefficient dropped by around 33%. Moreover, the sour mixed-gas permeation properties using a high H2S-content multicomponent gas mixture (20 vol% H2S) were measured for 6FDA-TPA and 6FDA-TPA(t-Bu) membranes at feed pressures up to 500 psi and 22 °C. The membranes showed nonideal behavior in the presence of hydrogen sulfide, where both membranes performed similarly and the effect of the bulky group (t-Bu) was negligible.
Hayek, A.; Alsamah, A.; Qasem, E. A.; Alaslai, N.; Alhajry, R. H.; Yahaya, G. O.; Separation and Purification Technology, 2019, 227, 115713. DOI: 10.1016/j.seppur.2019.115713.
14. Pure- and sour mixed-gas transport properties of 4,4'-methylenebis(2,6-diethylaniline) copolyimide membranes.
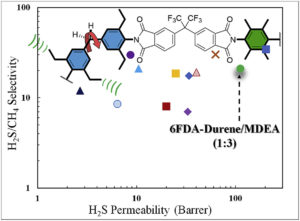 Three aromatic 6FDA-Durene/MDEA random copolyimides with different Durene:MDEA molar ratios (3:1; 1:1 and 1:3) were synthesized, and pure gas permeation properties through their dense films were investigated. Compared to their corresponding homopolymers (6FDA-MDEA and 6FDA-Durene), the copolymers exhibit significant improvement in gas separation performance, as the membranes display improved gas transport properties. For example, for pure gas, the CO2 permeability for 6FDA-Durene/MDEA (3:1) is around 234 Barrer and CO2/CH4 selectivity equal to 17.8 obtained at 22 °C and feed pressure of 100 psi. In addition, for sweet gas mixture consisting of 10, 59, 30 and 1 vol% CO2, CH4, N2 and C2H6, respectively, the 6FDA-Durene/MDEA (1:3) copolyimide membrane shows CO2 permeability of 73 Barrer and CO2/CH4 selectivity of about 24 at an elevated pressure of 800 psi. For a sour gas mixture with high H2S content (20 vol % H2S in the feed gas), the membrane exhibits H2S/CH4 selectivity of 21; and H2S permeability of 112 Barrer. The transport properties of the herein reported membrane are similar to or even better than the performance exhibited by some polymeric membranes reported in the literature.
Three aromatic 6FDA-Durene/MDEA random copolyimides with different Durene:MDEA molar ratios (3:1; 1:1 and 1:3) were synthesized, and pure gas permeation properties through their dense films were investigated. Compared to their corresponding homopolymers (6FDA-MDEA and 6FDA-Durene), the copolymers exhibit significant improvement in gas separation performance, as the membranes display improved gas transport properties. For example, for pure gas, the CO2 permeability for 6FDA-Durene/MDEA (3:1) is around 234 Barrer and CO2/CH4 selectivity equal to 17.8 obtained at 22 °C and feed pressure of 100 psi. In addition, for sweet gas mixture consisting of 10, 59, 30 and 1 vol% CO2, CH4, N2 and C2H6, respectively, the 6FDA-Durene/MDEA (1:3) copolyimide membrane shows CO2 permeability of 73 Barrer and CO2/CH4 selectivity of about 24 at an elevated pressure of 800 psi. For a sour gas mixture with high H2S content (20 vol % H2S in the feed gas), the membrane exhibits H2S/CH4 selectivity of 21; and H2S permeability of 112 Barrer. The transport properties of the herein reported membrane are similar to or even better than the performance exhibited by some polymeric membranes reported in the literature.
Hayek, A.; Yahaya, G. O.; Alsamah, A.; Alghannam, A. A.; Jutaily, S. A.; Mokhtari, I.; Polymer, 2019, 166, 184 –195. DOI: 10.1016/j.polymer.2019.01.056.
13. High pressure pure- and mixed sour gas transport properties of Cardo-Type block co-polyimide membranes.
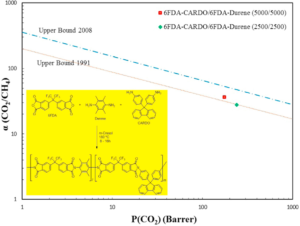
Multi-block co-polyimides 6FDA-CARDO/6FDA-Durene with varying segmental lengths were synthesized and pure and mixed sour gas transport properties through dense membranes were investigated as function of feed pressure, temperature and gas composition. Pure gas measurement indicated that block co-polyimide 6FDA-CARDO/6FDA-Durene (2500/2500) membrane exhibits high permeation properties and separation characteristics with CO2permeability and CO2/CH4 selectivity of up to 239 barrer and 28 respectively. The block co-polyimide also showed excellent performance under harsh sour gas environment (i.e. high H2S content of up to 36 vol% and feed pressure of up to 55 bar for a gas mixture consisting of CO2, CH4, N2, C2H6 and H2S). At 36 vol% H2S, the membrane exhibits H2S permeability and H2S/CH4 selectivity of up to 275 barrer and 24 respectively. These values and separation performance exhibited by the co-polyimide are comparable and very competitive even, as compared to the values obtained in some of the high performance polymeric membranes that have been reported in the literature. The stability of the block co-polyimide 6FDA-CARDO/6FDA-Durene membrane under these aggressive environments is quite impressive.
Alghannam, A. A.; Yahaya, G. O.; Hayek, A.; Mokhtari, I.; Saleem, Q.; Sewdan, D. A.; Bahamdan, A. A.; Journal of membrane science, 2018, 553, 32 – 42. DOI: 10.1016/j.memsci.2018.02.042.
12. A Green Technology for Fibre Modification, Part I: Attaching a Fluorescent Molecule to Cellulose Fibres and their Potential High-Value Applications.
 In this research, a bre modi cation technology has been developed based on the formation of an amide bond between the cellulose bre carboxyl group and a primary or secondary amino group of functional amino molecules. Using this technology, a uorescent molecule, 5-amino uorescein, has been bonded to commercial, bleached Kraft pulp bres in aqueous media under mild conditions. Papers made from a bre furnish containing as little as 0.005% 5-amino uorescein-bonded bres show a built-in security feature authenticable upon light excitation. The total chemical dose for making such papers is only 0.5 ppm on bres. The 5-amino uorescein-bonded bres can potentially be used to produce high-value papers with a built-in security feature such as passports and sports tickets. They may also be used to identify a bre furnish.
In this research, a bre modi cation technology has been developed based on the formation of an amide bond between the cellulose bre carboxyl group and a primary or secondary amino group of functional amino molecules. Using this technology, a uorescent molecule, 5-amino uorescein, has been bonded to commercial, bleached Kraft pulp bres in aqueous media under mild conditions. Papers made from a bre furnish containing as little as 0.005% 5-amino uorescein-bonded bres show a built-in security feature authenticable upon light excitation. The total chemical dose for making such papers is only 0.5 ppm on bres. The 5-amino uorescein-bonded bres can potentially be used to produce high-value papers with a built-in security feature such as passports and sports tickets. They may also be used to identify a bre furnish.
Hu, T. Q.; Hayek, A.; Journal of Science & Technology for Forest Products and Processes (J-FOR), 2013, VOL.3, NO.3, 34 – 40.
11. New insight in boron chemistry: Application in two-photon absorption.
 Two groups of one-dimensional (1D) boron containing two-photon absorbing fluorophores have been prepared and characterized. One group includes boron atoms incorporated in the conjugated or pseudo conjugated central core and the other contain a boron cluster as an acceptor group at one end of the fluorophores. Two boron containing central cores (with two boron atoms) have been explored: the cyclodiborazane and the pyrazabole moieties. The chosen boron cluster, p-carborane, contains 10 boron atoms. All the prepared fluorophores present high two-photon absorption cross-sections. Some water-soluble as well as lipophylic dyes have been prepared and used in bio-imaging.
Two groups of one-dimensional (1D) boron containing two-photon absorbing fluorophores have been prepared and characterized. One group includes boron atoms incorporated in the conjugated or pseudo conjugated central core and the other contain a boron cluster as an acceptor group at one end of the fluorophores. Two boron containing central cores (with two boron atoms) have been explored: the cyclodiborazane and the pyrazabole moieties. The chosen boron cluster, p-carborane, contains 10 boron atoms. All the prepared fluorophores present high two-photon absorption cross-sections. Some water-soluble as well as lipophylic dyes have been prepared and used in bio-imaging.
Bolze, F.; Hayek, A.; Sun, X.H.; Baldeck, P.L.; Bourgogne, C.; Nicoud, J.-F.; Optical Materials, 2011, 33(9), 1453– 1458. DOI: 10.1016/j.optmat.2011.02.035.
10. Boron Containing Two-Photon Absorbing Chromophores. 3. One- and Two-Photon Photophysical Properties of p- Carborane Containing Fluorescent Bioprocess.
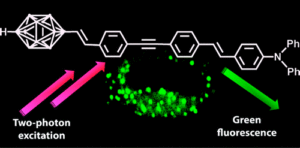 Boron-contg. two-photon-absorbing fluorophores have been prepd. as new bifunctional mols., potentially useful in two-photon excited microscopy (TPEM) and boron neutron capture therapy. They are based on a one-dimensional conjugated system contg. a p-carborane entity at one end of the mol. and various electron-donating groups contg. oxygen or nitrogen atoms at the other end. The authors investigated their one- and two-photon photophys. properties. They showed efficient fluorescence in an org. solvent, as well as in water for two of them, allowing microscopy on cell cultures. High two-photon absorption cross sections were detd. in the 700-900 nm range. TPEM images were obtained with these new p-carborane-contg. fluorophores, with laser intensities in the submilliwatt range.
Boron-contg. two-photon-absorbing fluorophores have been prepd. as new bifunctional mols., potentially useful in two-photon excited microscopy (TPEM) and boron neutron capture therapy. They are based on a one-dimensional conjugated system contg. a p-carborane entity at one end of the mol. and various electron-donating groups contg. oxygen or nitrogen atoms at the other end. The authors investigated their one- and two-photon photophys. properties. They showed efficient fluorescence in an org. solvent, as well as in water for two of them, allowing microscopy on cell cultures. High two-photon absorption cross sections were detd. in the 700-900 nm range. TPEM images were obtained with these new p-carborane-contg. fluorophores, with laser intensities in the submilliwatt range.
Nicoud, J.-F; Bolze, F.; Sun, X.-H.; Hayek, A.; Baldeck, P.; Inorg. Chem., 2011, 50(10), 4272 – 4278. DOI: 10.1021/ic102043v.
9. Boron Containing Two-Photon Absorbing Chromophores. 2. Fine Tuning of the One- and Two-Photon Photophysical Properties of Pyrazabole Based Fluorescent Bio-Probes.
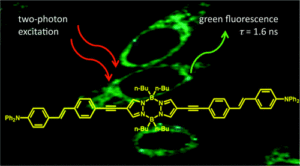 New boron containing two-photon absorbing fluorescent bioprobes built around a pyrazabole central core have been prepared. They bear various conjugated systems and end groups leading to efficient fluorescence in organic solvents. High two-photon absorption cross sections were determined in the 500−800 nm range that allow live-cell two-photon excited microscopy images to be obtained with laser intensities in the milliwatt range.
New boron containing two-photon absorbing fluorescent bioprobes built around a pyrazabole central core have been prepared. They bear various conjugated systems and end groups leading to efficient fluorescence in organic solvents. High two-photon absorption cross sections were determined in the 500−800 nm range that allow live-cell two-photon excited microscopy images to be obtained with laser intensities in the milliwatt range.
Hayek, A.; Bolze, F.; Bourgogne, C.; Baldeck, P. L.; Didier, P.; Arntz, Y.; Nicoud, J.-F.; Inorg. Chem., 2009, 48 (19), 9112 – 9119. DOI: 10.1021/ic900627g
8. Layer-By-Layer Dendritic Growth of Hyperbranched Thin Films for Surface Sol-Gel Syntheses of Conformal, Functional, Nanocrystalline Oxide Coatings on Complex 3-D (Bio)silica Templates.
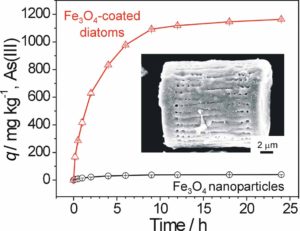
Here, a straightforward and general method for the rapid dendritic amplification of accessible surface functional groups on hydroxylated surfaces is described, with focus on its application to 3D biomineral surfaces. Reaction of hydroxyl-bearing silica surfaces with an aminosilane, followed by alternating exposure to a dipentaerythritol-derived polyacrylate solution and a polyamine solution, allows the rapid, layer-by-layer (LBL) build-up of hyperbranched polyamine/polyacrylate thin films. Characterization of such LBL-grown thin films by AFM, ellipsometry, XPS, and contact angle analyses reveals a stepwise and spatially homogeneous increase in film thickness with the number of applied layers. UV–Vis absorption analyses after fluorescein isothiocyanate labeling indicate that significant amine amplification is achieved after the deposition of only 2 layers with saturation achieved after 3–5 layers. Use of this thin-film surface amplification technique for hydroxyl-enrichment of biosilica templates facilitates the conformal surface sol–gel deposition of iron oxide that, upon controlled thermal treatment, is converted into a nanocrystalline (∼9.5 nm) magnetite (Fe3O4) coating. The specific adsorption of arsenic onto such magnetite-coated frustules from flowing, arsenic-bearing aqueous solutions is significantly higher than for commercial magnetite nanoparticles (≤50 nm in diameter).
Wang, G.; Fang, Y.; Kim, P.; Hayek, A.; Weatherspoon, M. R.; Perry, J. W.; Sandhage, K. H.; Marder, S. R.; Jones, S. C.; Adv. Funct. Mater., 2009, 19(17), 2768 – 2776. Cover Article. DOI: 10.1002/adfm.200900402.
7. Fabrication of Low-Shrinkage Microporous 3D Polymer Structures Through Interference Lithography.
 Multi-beam interference lithography offers an efficient route to fabricate various microporous three-dimensional (3D) structures for applications such as micro-optics, microfluidics, microelectro-mechanical systems, and optical data storage. However, the 3D structures fabricated by interference lithography could be distorted by the high refractive index of the photoresist film and the shrinkage of photoresist. For example, we found ~41% shrinkage in the [111] direction when fabricating diamond-like structures from the commercially available negative-tone photoresist, SU8. This large shrinkage together with refraction effect due to large refractive index of SU8 causes structure distortion and residual strain in the 3D porous structures. In order to minimize the structure distortion, we have synthesized narrowly dispersed poly(glycidyl methacrylate) (PGMA) with controlled number of epoxy groups by atom transfer radical polymerization. In contrast to SU8, the 3D structures fabricated from PGMA show a much lower volumetric shrinkage, 5.9%, and fewer cracks in the film.
Multi-beam interference lithography offers an efficient route to fabricate various microporous three-dimensional (3D) structures for applications such as micro-optics, microfluidics, microelectro-mechanical systems, and optical data storage. However, the 3D structures fabricated by interference lithography could be distorted by the high refractive index of the photoresist film and the shrinkage of photoresist. For example, we found ~41% shrinkage in the [111] direction when fabricating diamond-like structures from the commercially available negative-tone photoresist, SU8. This large shrinkage together with refraction effect due to large refractive index of SU8 causes structure distortion and residual strain in the 3D porous structures. In order to minimize the structure distortion, we have synthesized narrowly dispersed poly(glycidyl methacrylate) (PGMA) with controlled number of epoxy groups by atom transfer radical polymerization. In contrast to SU8, the 3D structures fabricated from PGMA show a much lower volumetric shrinkage, 5.9%, and fewer cracks in the film.
Xu, Y.; Hayek, A.; Zhu, X.; Okada, T.; Moon, J. H.; Barlow, S.; Yang, S.; Marder, S. R.; Polymer Preprints, 2008, 49(2), 529-530.
6. Poly(Glycidyl Methacrylate)S with Controlled Molecular Weights as Low-Shrinkage Resins for 3D Multibeam Interference Lithography.
 Poly(glycidyl methacrylate) has been shown to be a useful material for fabrication of photonic crystal templates using multibeam interference lithography, since it exhibits lower shrinkage than conventional SU8.
Poly(glycidyl methacrylate) has been shown to be a useful material for fabrication of photonic crystal templates using multibeam interference lithography, since it exhibits lower shrinkage than conventional SU8.
Hayek, A.; Xu, Y.; Okada, T.; Barlow, S.; Zhu, X.; Moon, J. H.; Marder, S. R.; Yang, S.; J. Mater. Chem., 2008, 18, 3316-3318. DOI: 10.1039/b809656b.
5. Cell-Permeant Cytoplasmic Blue Fluorophores Optimized for In Vivo Two-Photon Microscopy with Low-Power Excitation.
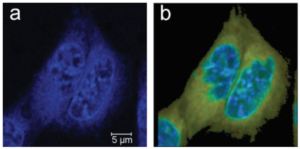 Because of the spreading of nonlinear microscopies in biology, there is a strong demand for specifically engineered probes in these applications. Herein, we report on the imaging properties in living cells and nude mice brains of recently developed water soluble blue fluorophores that show efficient diffusion through cell membranes and blood–brain barriers. They are characterized by two-photon absorption cross-sections of 100–150 Goeppert-Mayer range in the near IR and fluorescence efficiencies of up to 72% in water. They were found to stain homogeneously the cytoplasm of cultured living cells within minutes. Moreover, their diffusion times and fluorescence characteristics in the cytoplasm suggest a hydrophobic association with intracellular membranes. Their intracellular fluorescent decays were found to be almost mono-exponential, a very favorable feature for fluorescence lifetime imaging. Two photon images of living cells were obtained with a good signal to noise ratio using laser powers in the sub-milliwatt range. This allows continuous imaging without significant photobleaching for tens of minutes. In addition, these fluorophores allowed in vivo three-dimensional two-photon imaging of mice cortex vasculatures and extra vasculature structures, with no sign of toxicity.
Because of the spreading of nonlinear microscopies in biology, there is a strong demand for specifically engineered probes in these applications. Herein, we report on the imaging properties in living cells and nude mice brains of recently developed water soluble blue fluorophores that show efficient diffusion through cell membranes and blood–brain barriers. They are characterized by two-photon absorption cross-sections of 100–150 Goeppert-Mayer range in the near IR and fluorescence efficiencies of up to 72% in water. They were found to stain homogeneously the cytoplasm of cultured living cells within minutes. Moreover, their diffusion times and fluorescence characteristics in the cytoplasm suggest a hydrophobic association with intracellular membranes. Their intracellular fluorescent decays were found to be almost mono-exponential, a very favorable feature for fluorescence lifetime imaging. Two photon images of living cells were obtained with a good signal to noise ratio using laser powers in the sub-milliwatt range. This allows continuous imaging without significant photobleaching for tens of minutes. In addition, these fluorophores allowed in vivo three-dimensional two-photon imaging of mice cortex vasculatures and extra vasculature structures, with no sign of toxicity.
Hayek, A.; Grichine, A.; Huault, T.; Ricard, C.; Bolze, F.; van der Sanden, B.; Vial, J.-C.; Mély, Y.; Duperray, A.; Baldeck, P. L.; Nicoud, J.-F.; Microscopy Research and Technique, 2007, 70, 880-885. DOI 10.1002/jemt.20493.
4. Conjugation of New Two-Photon Fluorophore to Polyethylenimine for Gene Delivery Imaging.
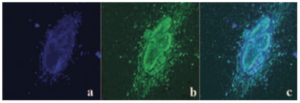
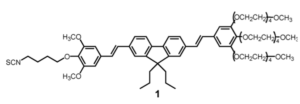 We report herein the molecular engineering of an efficient two-photon absorbing (TPA) chromophore based on a donor−donor bis-stilbenyl entity to allow conjugation with biologically relevant molecules. The dye has been functionalized using an isothiocyanate moiety to conjugate it with the amine functions of poly(ethylenimine) (PEI), which is a cationic polymer commonly used for nonviral gene delivery. Upon conjugation, the basic architecture and photophysical properties of the active TPA chromophore remain unchanged. At the usual N/P ratio (ratio of the PEI positive charges to the DNA negative charges) of 10 used for transfection, the transfection efficiency and cytotoxicity of the labeled PEI/DNA complexes were found to be comparable to those of the unlabeled PEI/DNA complexes. Moreover, when used in combination with unlabeled PEI (at a ratio of 1 labeled PEI to 3 unlabeled PEI), the labeled PEI does not affect the size of the complexes with DNA. The labeled PEI was successfully used in two-photon fluorescence correlation spectroscopy measurements, showing that at N/P = 10 most PEI molecules are free and the diffusion coefficient of the complexes is consistent with the 360 nm size measured by quasielastic light scattering. Finally, two-photon images of the labeled PEI/DNA complexes confirmed that the complexes enter into the cytoplasm of HeLa cells by endocytosis and hardly escape from the endosomes. As a consequence, the functionalized TPA chromophore appears to be an adequate tool to label the numerous polyamines used in nonviral gene delivery and characterize their complexes with DNA in two-photon applications.
We report herein the molecular engineering of an efficient two-photon absorbing (TPA) chromophore based on a donor−donor bis-stilbenyl entity to allow conjugation with biologically relevant molecules. The dye has been functionalized using an isothiocyanate moiety to conjugate it with the amine functions of poly(ethylenimine) (PEI), which is a cationic polymer commonly used for nonviral gene delivery. Upon conjugation, the basic architecture and photophysical properties of the active TPA chromophore remain unchanged. At the usual N/P ratio (ratio of the PEI positive charges to the DNA negative charges) of 10 used for transfection, the transfection efficiency and cytotoxicity of the labeled PEI/DNA complexes were found to be comparable to those of the unlabeled PEI/DNA complexes. Moreover, when used in combination with unlabeled PEI (at a ratio of 1 labeled PEI to 3 unlabeled PEI), the labeled PEI does not affect the size of the complexes with DNA. The labeled PEI was successfully used in two-photon fluorescence correlation spectroscopy measurements, showing that at N/P = 10 most PEI molecules are free and the diffusion coefficient of the complexes is consistent with the 360 nm size measured by quasielastic light scattering. Finally, two-photon images of the labeled PEI/DNA complexes confirmed that the complexes enter into the cytoplasm of HeLa cells by endocytosis and hardly escape from the endosomes. As a consequence, the functionalized TPA chromophore appears to be an adequate tool to label the numerous polyamines used in nonviral gene delivery and characterize their complexes with DNA in two-photon applications.
Hayek, A.; Ercelen, S.; Zhang, X.; Bolze, F.; Nicoud, J.-F.; Schaub, E.; Baldeck, P.-L.; Mély, Y.; Bioconjugate Chemistry, 2007, 18(3), 844-851. DOI 10.1021/bc060362h.
3. Two-Photon Water-Soluble Dyes and Their Amine-Reactive Derivatives for Two-Photon Bio-Imaging Applications.
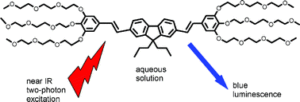
 Two series of water-soluble blue fluorescent two-photon excited chromophores for bio-imaging based on bis-stilbenic structure were synthesized.Wadsworth-Emmons reaction was used to build one dimensional D-p-aromatic core-p-D structures. The water solubility is induced by three oligo-ethylene glycol moieties in positions 3, 4, and 5 of the peripheral phenyl ring. Their two-photon absorption (TPA) cross-section in the range of 150 GM at 700 nm have been measured by the way of their two-photon excited fluorescence (TPEF) properties. It is 10 to 100 times higher than commercial dyes commonly used in bio-imaging. This makes these new TPA chromophores good candidates for in-vivo two-photon excited (TPE) microscopy.
Two series of water-soluble blue fluorescent two-photon excited chromophores for bio-imaging based on bis-stilbenic structure were synthesized.Wadsworth-Emmons reaction was used to build one dimensional D-p-aromatic core-p-D structures. The water solubility is induced by three oligo-ethylene glycol moieties in positions 3, 4, and 5 of the peripheral phenyl ring. Their two-photon absorption (TPA) cross-section in the range of 150 GM at 700 nm have been measured by the way of their two-photon excited fluorescence (TPEF) properties. It is 10 to 100 times higher than commercial dyes commonly used in bio-imaging. This makes these new TPA chromophores good candidates for in-vivo two-photon excited (TPE) microscopy.
Hayek, A.; Bolze, F.; Nicoud, J.-F.; Duperray, A.; Grichine, A.; Baldeck, P. L.; Vial, J.-C.; Nonlinear Optics, Quantum Optics, 2006, 35(1-3), 155-164.
2. Boron Containing Two-photon Absorbing Chromophores: Electronic Interaction through the Cyclodiborazane Core.
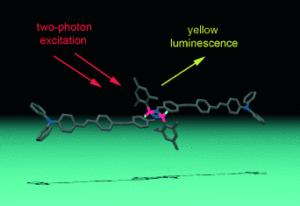 Through-bond coupling across a cyclodiborazane core results in large absorption cross sections in a new type of boron-containing two-photon-absorbing chromophore (see picture; B pink, N blue, C gray, H other than BH (white) omitted). This is a first step in the design of new dual-property chromophores for applications in biological science or medicine (e.g., two-photon imaging and boron neutron capture therapy).
Through-bond coupling across a cyclodiborazane core results in large absorption cross sections in a new type of boron-containing two-photon-absorbing chromophore (see picture; B pink, N blue, C gray, H other than BH (white) omitted). This is a first step in the design of new dual-property chromophores for applications in biological science or medicine (e.g., two-photon imaging and boron neutron capture therapy).
Hayek, A.; Nicoud, J.-F.; Bolze, F.; Bourgogne, C.; Baldeck, P. L.; Angew. Chem., Int. Ed. Engl., 2006, 45, 6466- 6469. DOI : 10.1002/anie.200602266.
1. Synthesis and Characterization of Water-Soluble Two-Photon Excited Blue Fluorescent Chromophores for Bioimaging.
 We report here the synthesis and characterization of a new type of non-ionic blue fluorescent water-soluble chromophores specifically designed for two-photon absorption microscopy. The water solubility is induced by introduction of short oligo(ethylene glycol) monomethyl ether moieties. This work has led to low molecular weight dyes with efficient two-photon absorption cross sections and high fluorescence quantum yield in organic solvents as well as in aqueous solutions.
We report here the synthesis and characterization of a new type of non-ionic blue fluorescent water-soluble chromophores specifically designed for two-photon absorption microscopy. The water solubility is induced by introduction of short oligo(ethylene glycol) monomethyl ether moieties. This work has led to low molecular weight dyes with efficient two-photon absorption cross sections and high fluorescence quantum yield in organic solvents as well as in aqueous solutions.
Hayek, A.; Bolze, F.; Nicoud, J.-F.; Baldeck, P.-L.; Mély, Y.; Photochem. Photobiol. Sci., 2006, 5, 102-106. DOI: 10.1039/b509843b.
